Clinical Trials for Head and Neck Cancer Patients
What Are Clinical Trials?
Clinical trials are research studies that involve patients. Through clinical trials, doctors and researchers find new ways to improve treatments and the quality of life for people with diseases.
Cancer clinical trials are designed to test new ways to:
- Treat Cancer
- Find and diagnose cancer
- Prevent cancer
- Manage symptoms of cancer; side effects from its treatment

Through clinical trials, doctors determine if new treatments are safe and effective, and whether or not they produce better outcomes than current treatments. Clinical trials also help find new ways to prevent and detect cancer. And they help improve the quality of life for people during and after treatment.
If you or your loved one have been diagnosed with head or neck cancer and need treatment, clinical trials are an option to consider. Trials are available for all stages of cancer.
How to Locate Head and Neck Clinical Trials
There is a substantial amount of head and neck cancer clinical trials awarded by government funding sources, as well as industry and pharmaceutical companies. It is important for patients to discuss clinical trials with their healthcare providers.
HNCA recommends researching head and neck cancer clinical trials either by going to www.ClinicalTrials.gov — a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world – or using our Clinical Trial Finder which is designed to be user-friendly for patients. HNCA also have some active clinical trials from ClinicalTrials.gov listed on our webpage here.

In addition, work with your healthcare provider on specific terminology as it relates to your or your loved one’s head and neck cancer diagnosis. For additional information on clinical trials, HNCA's webinar, "Exploring Clinical Trials in the Head and Neck Cancer" or the National Institutes of Health are some helpful resources.
Key information to know for your head and neck cancer clinical trial search includes:
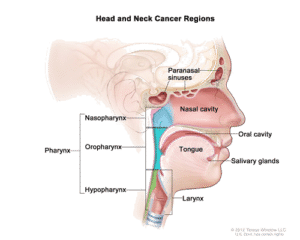
- Histology (squamous cell carcinoma, adenoid cystic carcinoma, adenocarcinoma, etc.)
- Site of head and neck cancer (tonsil, parotid gland, larynx, etc.)
- Recurrent or First Presentation. If recurrent, know how it was treated before (surgery, radiation, chemotherapy, and drugs used for treatment).
- HPV-related
Clinical Trials Phases*
Clinical trials involving new drugs are commonly classified into five phases. Each phase of the drug approval process is treated as a separate clinical trial. The drug-development process will normally proceed through four phases over many years.
If the drug successfully passes through phases 0, 1, 2, and 3, it will usually be approved by the national regulatory authority for use in the general population. Before pharmaceutical companies start clinical trials on a drug, they will also have conducted extensive preclinical studies. Each phase has a different purpose and helps scientists answer a different question.
| Phase | Aim | Notes |
| Phase 0 | Pharmacodynamics & pharmacokinetics in humans |
Phase 0 trials are the first-in-human trials. Single subtherapeutic doses of the study drug or treatment are given to a small number of subjects (10 to 15) to gather preliminary data on the agent's pharmacodynamics (what the drug does to the body) and pharmacokinetics (what the body does to the drugs).[32] For a test drug, the trial documents the absorption, distribution, metabolization, and removal (excretion) of the drug, and the drug's interactions within the body, to confirm that these appear to be as expected. |
| Phase 1 | Screening for safety. |
Testing within a small group of people (20–80) to evaluate safety, determine safe dosage ranges, and begin to identify side effects. A drug's side effects could be subtle or long term, or may only happen with a few people, so phase 1 trials are not expected to identify all side effects. |
| Phase 2 | Establishing the efficacy of the drug, usually against a placebo. |
Testing with a larger group of people (100–300) to determine efficacy and to further evaluate its safety. The gradual increase in test group size allows for the evocation of less-common side effects. |
| Phase 3 | Final confirmation of safety and efficacy. | Testing with large groups of people (1,000–3,000) to confirm its efficacy, evaluate its effectiveness, monitor side effects, compare it to commonly used treatments, and collect information that will allow it to be used safely. |
| Phase 4 | Safety studies during sales. | Postmarketing studies delineate additional information, including the treatment's risks, benefits, and optimal use. As such, they are ongoing during the drug's lifetime of active medical use. (Particularly relevant after approval under FDA Accelerated Approval Program) |
*This article uses material from the Wikipedia article https://en.wikipedia.org/wiki/Clinical_trial which is released under the Creative Commons Attribution-ShareAlike 3.0 Unported License.

